CUHK develops an interventional catheterisation-integrated swarming microrobotic platform for aneurysm embolisation treatment: a new non-invasive approach
- The swarming self-adhesive microgels used in the new platform are more controllable than the existing materials for embolisation of aneurysms, overcoming the bottleneck of complete filling of aneurysms due to high-speed blood flow, and reducing complications.
- Image-guided interventional delivery is also used in this new strategy for in situ tracking and performance evaluation inside biological tissues.
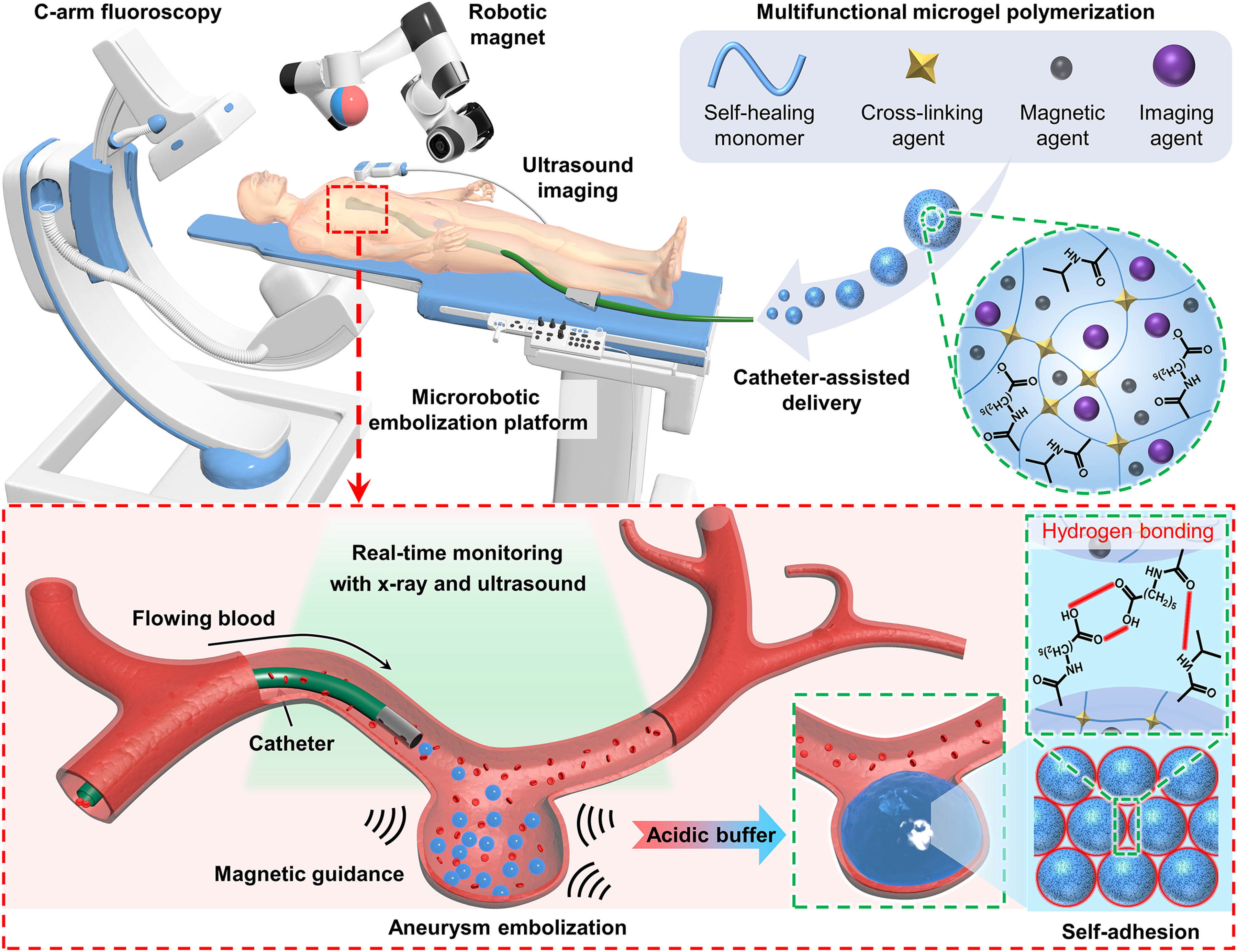
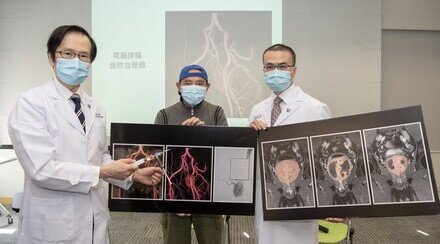
An interventional catheterisation-integrated swarming microrobotic platform for real-time medical imaging guided embolisation of aneurysm,aiming to address the bottlenecks of conventional embolisation therapy in filling and targeting efficiencies. Each building block of the microrobotic swarm is a micro-sized sphere composed of pH-responsive self-healing hydrogel matrix, magnetic nanoparticles and imaging dopants.
A collaborative research team led by Professor Zhang Li from the Faculty of Engineering at The Chinese University of Hong Kong (CUHK) and Professor Simon Yu Chun-ho from CUHK’s Faculty of Medicine (CU Medicine) has developed swarming self-adhesive microgels for a new embolisation strategy to prevent aneurysms from continuously bulging to a point of rupture. This collaborative work was published recently in the renowned international research journal Science Advances.
The microrobotic embolisation platform designed by the research team has overcome the bottleneck in aneurysm complete filling under highly dynamic blood flow and provides an image-guided interventional delivery strategy for in situ tracking and performance evaluation inside biological tissues. It is also potentially applicable to non-invasive embolisation treatments of other organs for bleeding control.
About aneurysm and the limitations of current therapeutic methods
Aneurysm, a typical vascular malformation, refers to an abnormal, outward bulging or ballooning area in the blood vessel wall. It can occur in many parts of the human body without any symptoms, including the aorta, abdomen and brain, but can also induce severe internal bleeding if it ruptures and become lethal, with a mortality rate of up to 40% in some parts of the body.
Although current embolisation treatment based on the endovascular delivery of embolic agents, such as metallic coils, polymeric spheres or a gelling solution, provides a clinically feasible means to block the aneurysm sac and stop blood from entering into an aneurysm, embolisation can fail to completely block an aneurysm sac and it can lead to serious complications, such as unintentional occlusion of distal blood vessels by the overflow or fragmentation of embolic materials, especially for giant aneurysms or an aortic dissection that takes place in larger vascular spaces. There is therefore an urgent need for a new embolisation strategy with better filling controllability, endovascular environment compatibility and clinical performance.
The new platform overcomes the bottleneck of complete filling of aneurysms due to high-speed blood flow
Each building block of the microswarm used in the newly developed swarming microrobotic platform is a micro-sized sphere composed of a pH-responsive, self-healing hydrogel matrix1, magnetic nanoparticles and imaging dopants. Assisted by a catheter and under real-time image guidance, swarming microgels are deployed and concentrated into the aneurysm sac. The addition of an acidic solution activates the self-adhesive mechanism among the microgels, welding them into a single entity (see details in appendix).
The team has achieved efficient, selective accumulation of microgels in an aneurysm model and the human placenta with a filling ratio of more than 95% in an artificial blood flow environment with a mean velocity of up to 20 cm/s. The microrobotic embolus exhibits strong and stable self-adhesion in a physiological environment for at least half a year, as well as satisfactory bio- and hemo-compatibility.
Professor Zhang Li from the Department of Mechanical and Automation Engineering at CUHK’s Faculty of Engineering said, “In this collaborative research work with CU Medicine, we demonstrated a new approach using an image-guided microrobotic platform with magnetic microgels for endovascular embolisation. In the future, we will keep conducting research that furthers our fundamental understanding of micro- and nanorobot swarms with better in vivo imaging and remote control. We will also continue with our joint efforts with our medical school colleagues to translate our research on miniature medical robots into real-world clinical applications.”
Professor Simon Yu Chun-ho, Professor in CU Medicine’s Department of Imaging and Interventional Radiology, commented, “This new embolisation technology is a good illustration of the value of collaborative research between medicine and engineering, from addressing a clinical problem to formulating feasible, biocompatible concepts to validating its clinical safety and efficacy. I can see great potential for further development and application of magnetic and image-guided microrobots in the field of endovascular interventional treatments.”
The research team is now working together closely to promote the development of the microrobotic embolisation platform via animal study and to conduct further preclinical studies and clinical evaluations. The team envisions that the development of the novel embolisation therapy will provide a non-invasive approach for efficient, safe, personalised treatment of various aneurysm-related diseases.
This work is supported by the Hong Kong Research Grants Council (RGC), National Natural Science Foundation of China, Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences (CAS) – CUHK Joint Laboratory of Robotics and Intelligent Systems, and Multi-Scale Medical Robotics Center under InnoHK at the Hong Kong Science Park.
Note 1: pH-responsive, self-healing hydrogel matrix is a substance that can self-adhere in response to changes in pH, such as exposure to acidic solutions.
Appendix
How does the newly developed swarming microrobotic platform work?
- It is initiated by catheter-assisted delivery and the deployment of swarming microgels to the aneurysm neck, which provides a highway across biological barriers and protects microrobots from the immune system.
- A programmed external magnetic field is applied to navigate and concentrate swarming microrobots into the aneurysm sac under the real-time guidance of ultrasound and fluoroscopy imaging.
- An acidic buffer solution is injected via a catheter to activate a self-adhesive mechanism among the microgels, welding them into a single entity.
- The catheter and magnetic field are removed.
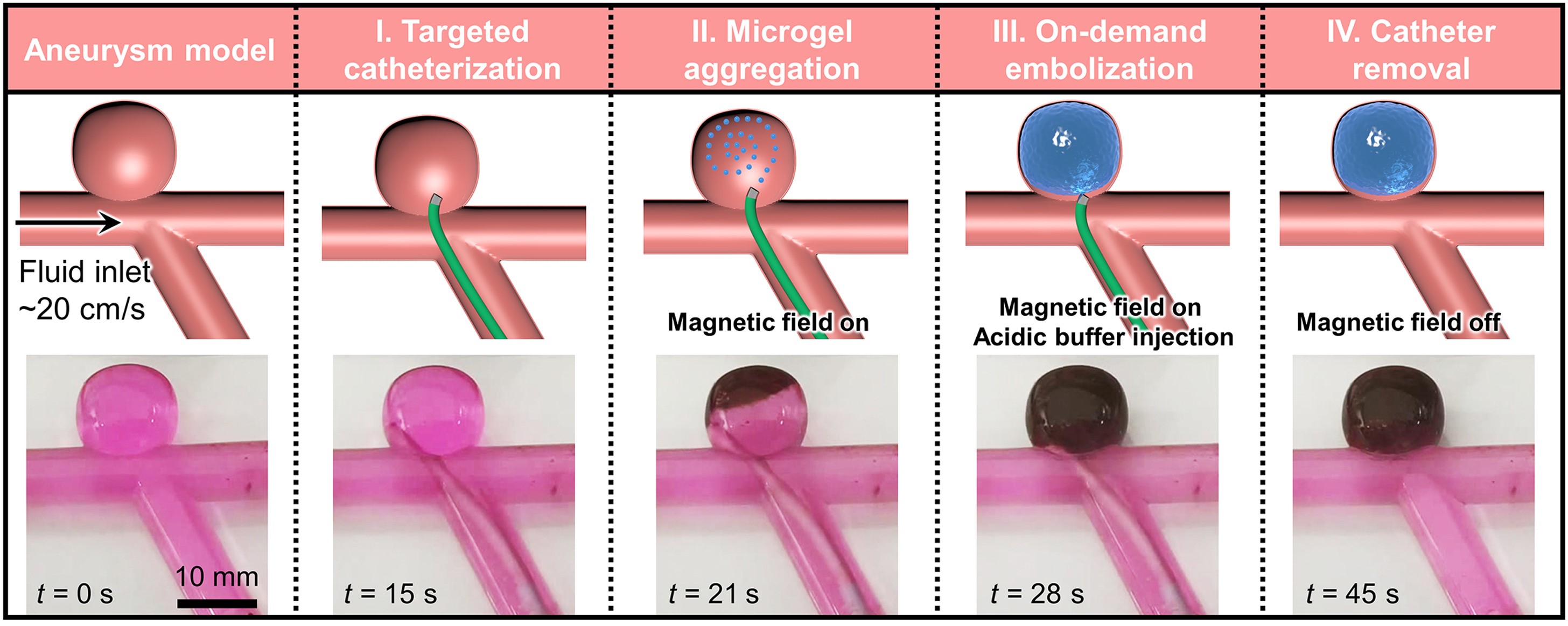
The schematic and experimental results of swarming self-adhesive microgels enabled on-demand embolisation in aneurysm model, including targeted catheterisation (I), deployment and active accumulation of swarming microgels into aneurysm sac under the actuation of robotic magnet (II), on-demand embolisation via mild acid stimulus (III), and the removal of catheter and robotic magnet (IV).